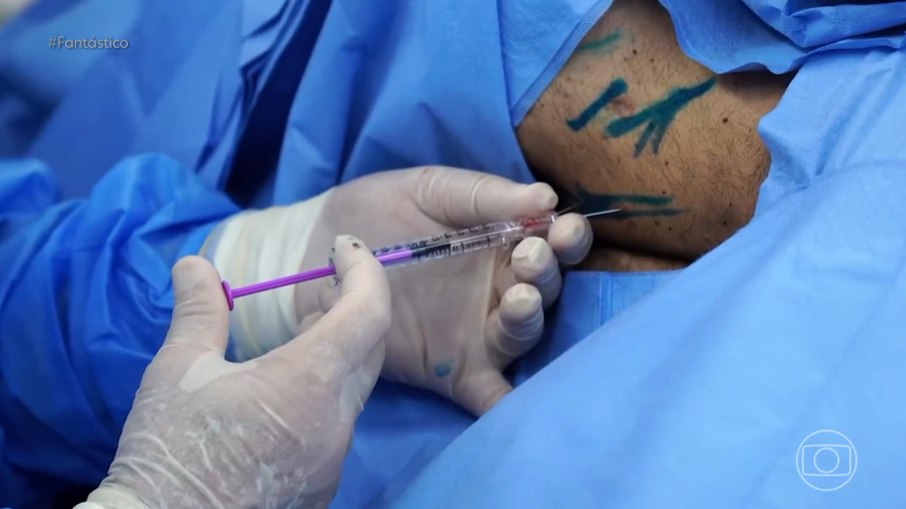
Accessing Polylaminin in Brazil requires navigating a specific intersection of ANVISA regulations, clinical trial protocols, and constitutional health rights.
While primarily discussed within the context of Phase I clinical trials (the 72-hour acute window), international patients may access this neuro-regenerative therapy through Compassionate Use (Uso Compassivo) or judicial injunctions (Tutelas de Urgência) for chronic spinal cord injuries.
What is Polylaminin and Why is Brazil the Epicenter of this Research?
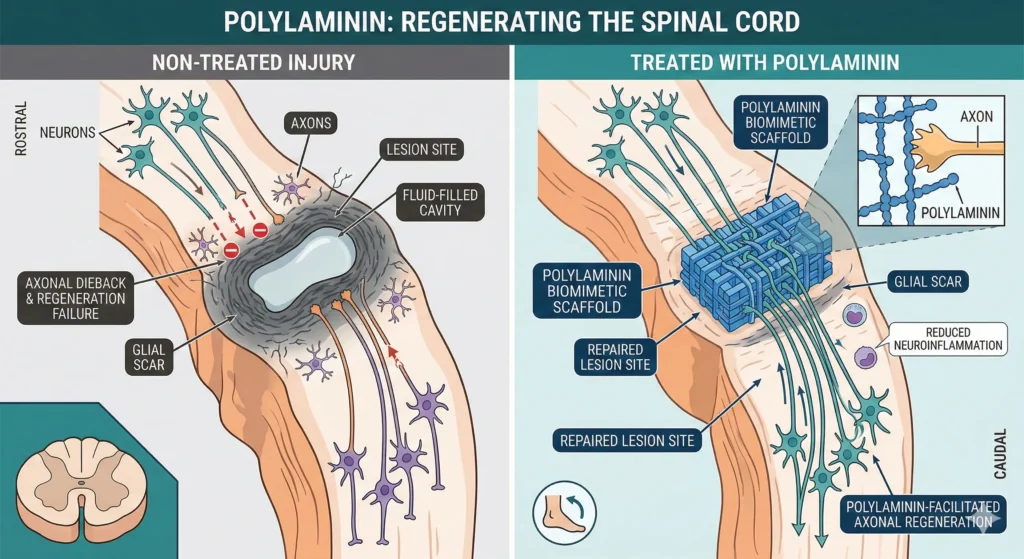
Polylaminin is a synthetic protein developed to mimic the natural extracellular matrix, specifically engineered to promote neuro-regeneration and bridge the gap in spinal cord injuries (SCI).
Unlike traditional treatments that focus only on stabilizing the spine, this Brazilian-patented biotechnology aims to create a permissive environment for axonal regrowth, effectively “reconnecting” the nervous system.
Brazil has become the global focal point for this therapy due to the groundbreaking partnership between the Federal University of Rio de Janeiro (UFRJ) and the Cristália Laboratory.
For an international patient, it is crucial to understand that while the drug is technically “experimental,” the Brazilian Federal Constitution treats the Right to Health (Article 196) as an absolute priority. This creates a unique legal landscape where even non-residents can, under specific legal frameworks, petition the government or private entities for access to cutting-edge treatments that are not yet available in the US, Europe, or Asia.
Micro-Scenario: > Imagine a patient from the United States who suffered a T10 injury two years ago. In most jurisdictions, they are told there are “no further options.” In Brazil, however, a specialized attorney can use the Polylaminin clinical data to argue that the patient has a legal right to “Compassionate Use,” bypassing the acute window required by the ongoing clinical trials.
The Brazilian Healthcare System (SUS) and the Cost Reality for Citizens
The Brazilian Unified Health System (SUS) is built on the principle of universal access. Once a drug like Polylaminin is fully approved by ANVISA and incorporated by CONITEC, it must be provided free of charge to any Brazilian citizen.
Under the current experimental protocols and agreements between researchers and the government, the commitment is that no Brazilian patient will pay for the substance itself during the clinical trial or after its potential inclusion in the public system.
For Brazilians, the financial burden is virtually zero regarding the medication, as the development was funded by public resources (UFRJ) and a national laboratory (Cristália).
The only current “costs” involve hospital stays or specialized surgical teams if the patient opts for a private hospital rather than a SUS-affiliated research center.
For an international patient, this creates a stark contrast: while the medicine is low-cost to produce, the legal and logistical infrastructure to access it as a non-resident is where the primary investment lies.
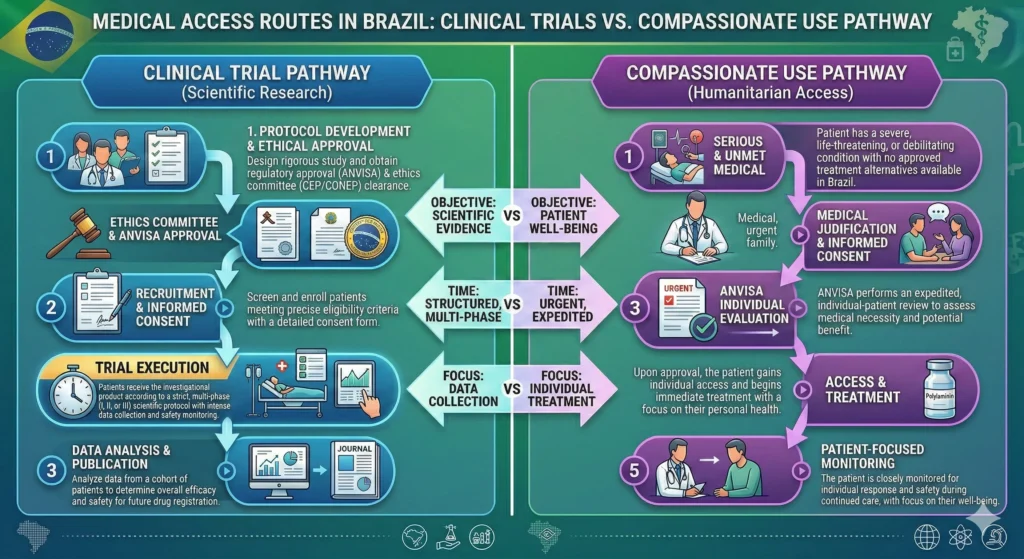
Clinical Trial Eligibility vs. The Freedom of Compassionate Use
A common misconception is that a patient must wait for a clinical trial to fail or a specific window to close before seeking help. In the Brazilian legal and regulatory framework, Compassionate Use (Uso Compassivo) is not a “second option,” but a primary legal pathway for those who do not fit the strict eligibility criteria of the ongoing Phase I study.
Clinical trials are designed for scientific validation and have extremely narrow profiles (often excluding patients with certain pre-existing conditions, specific age groups, or injury types). If you are ineligible for the trial for any reason, whether it is the time since the injury (post-72 hours), your medical history, or simply because the trial’s limited spots are filled, the law still protects your right to try.
The ANVISA RDC 38/2013 serves as a “regulatory bridge.” It allows the use of Polylaminin for any patient with a serious, debilitating condition where no satisfactory alternative exists in the country. This means that even if the “72-hour trial” is active, you can simultaneously petition for compassionate access based on your unique clinical necessity.
The Role of the Brazilian Judiciary: Securing the Right to Experimental Treatment
In Brazil, the “Right to Life” and “Dignity of the Human Person” are supreme legal principles. When an international patient seeks Polylaminin, the Judiciary acts as the ultimate guarantor.
If the laboratory or the regulatory agency creates bureaucratic hurdles, a specialized attorney can file an Injunction (Tutela de Urgência). To the Brazilian courts, “experimental” does not mean “forbidden.” If there is enough scientific evidence (like the UFRJ papers and Phase I safety data) showing that the drug is safe and potentially effective, judges often grant the right to access.
Key Legal Arguments used in these cases:
- Irreparable Harm: Every day without neuro-regenerative stimulus is a day of potential permanent loss.
- Scientific Grounding: Polylaminin is not “alternative medicine”; it is a patented biotechnology with peer-reviewed backing.
- Universal Health Right: The Brazilian Constitution does not discriminate based on nationality when a life or a fundamental bodily function is at stake.
Historical Precedents: How Foreigners and “Experimental” Claims Win in Brazil
The Brazilian legal system is historically one of the most receptive in the world regarding the Judicialization of Health. To understand why an international patient has a high chance of success with Polylaminin, we must look at how the Supreme Court (STF) and Federal Courts have handled similar cases.
1. The Polylaminin Precedents (2026)
As of early 2026, Brazilian courts have already begun setting a specific precedent for Polylaminin. In landmark cases, patients have secured access to the substance even before the Phase I trials were fully operational for their specific injury type. The courts ruled that the Compassionate Use framework (RDC 38/2013) allowed for an exception to the general rule that the State does not provide unregistered drugs. The judges noted that because the laboratory (Cristália) was willing to provide the drug and the scientific safety data was already robust, the patient’s right to hope outweighed regulatory bureaucracy.
2. The “Right to Try” Doctrine
A crucial legal concept in Brazil is that the Judiciary will intervene when the regulatory agency (ANVISA) is perceived as too slow to respond to terminal or irreversible conditions. Polylaminin has solid academic backing from the UFRJ, making it much easier for a judge to grant a “Right to Try” injunction compared to other less-studied substances.
3. Foreigners’ Standing in Health Lawsuits
Historical precedents from the STF (Supreme Federal Court), such as the judgment of RE 612.043, affirm that the right to health is a universal human right. Brazilian jurisprudence consistently rules that anyone on national territory—regardless of nationality or residency—must have their life and bodily integrity protected. If the treatment is available in Brazil and is the only hope for a patient’s recovery, the courts have historically granted access to foreigners.
Logistics for International Patients: The Roadmap to Brazil
Coming to Brazil for treatment is more than just a flight; it is a coordinated legal-medical operation. Here is what is required:
- Medical Records Translation: All your SCI history must be translated into Portuguese by a sworn translator (Tradução Juramentada).
- Local Medical Sponsorship: You must be linked to a Brazilian neurosurgeon or neurologist who will be the “Responsible Physician” for the application.
- Legal Power of Attorney: You will need to grant a Brazilian lawyer the power to represent you before the courts and ANVISA.
- The “Cristália” Connection: Your legal team will coordinate with the laboratory to ensure the logistics of the dose (which must be kept in specific conditions) are synchronized with your surgery date in a Brazilian hospital.
Accessing Polylaminin in Brazil is not a simple transaction; it is a specialized legal procedure. However, the combination of low production costs, a pro-patient Judiciary, and a robust Compassionate Use framework makes Brazil the most viable destination in the world for those seeking spinal cord regeneration today.
Polylaminin in Brazil: FAQ for International Patients
1. What is the legal status of Polylaminin in Brazil as of 2026?
Polylaminin is currently categorized as an experimental drug under clinical investigation (Phase I/II). While it does not yet have a definitive commercial registration (sanitary registry) for over-the-counter sale, it is legally accessible through two regulated pathways: authorized clinical trials and the Compassionate Use program regulated by ANVISA.
2. Can a non-resident foreigner apply for Compassionate Use?
Yes. The Brazilian Constitution and the Migration Law (Law 13.445/2017) guarantee health rights to anyone on Brazilian soil. There is no nationality restriction for requesting humanitarian access to experimental treatments, provided the legal and medical requirements are met.
3. Do I need to be in Brazil to start the legal process?
No. You can hire a Brazilian Health Attorney and provide a Power of Attorney (Procuração) from your home country (usually apostilled or via consulate). The legal suit or the ANVISA application can start while you are preparing your travel logistics.
4. What is the “72-hour window” mentioned in clinical trials?
The 72-hour window is a scientific requirement for the Phase I Clinical Trial focused on acute injuries. It aims to test the protein’s efficacy in preventing initial scar tissue. However, this window does not apply to Compassionate Use, which can be requested for chronic cases.
5. Is Polylaminin effective for chronic spinal cord injuries (years after the accident)?
While the most publicized trials focus on acute cases, the biological premise of Polylaminin—providing a scaffold for axonal regrowth—is theoretically applicable to chronic injuries. In these cases, the surgery may involve clearing existing scar tissue (fibrosis) before applying the protein.
6. How much does the medication cost for a foreigner?
A foreigner typically pays for the “Medical-Legal Package.” This includes hospital fees, the surgical team, and legal representation.
7. Does the Brazilian government (SUS) pay for a foreigner’s treatment?
Generally, no. While SUS provides the medication for free to citizens, foreigners usually access it via the private healthcare network or by paying the laboratory directly after a court order. However, the right to access the medication is guaranteed.
8. How long does a “Judicial Injunction” (Liminar) take to be granted?
In urgent health cases involving spinal cord regeneration, a Brazilian judge can issue a “Tutela de Urgência” within 24 to 72 hours after the lawsuit is filed.
9. What medical documents do I need from my home country?
You need your full clinical history, specifically:
Original MRI/CT scans (DICOM files).
Detailed surgical reports from the time of injury.
A “Statement of Therapeutic Failure” from your doctor, confirming no other options are available in your country.
10. Does the documentation need to be translated?
Yes. All foreign documents must undergo a Sworn Translation (Tradução Juramentada) into Portuguese to be valid before Brazilian courts and ANVISA.
11. Which laboratory produces Polylaminin?
The production is handled by Cristália, one of Brazil’s most advanced pharmaceutical laboratories, in partnership with researchers from the Federal University of Rio de Janeiro (UFRJ).
12. Are there known side effects?
Phase I trials focus precisely on safety. So far, Polylaminin has shown high biocompatibility because it mimics natural proteins (laminin) found in the human body, minimizing the risk of rejection.
13. Can I choose any hospital in Brazil for the procedure?
The procedure requires a high-complexity neurosurgical center. Most cases are handled in São Paulo or Rio de Janeiro, where the specialized teams familiar with the Polylaminin protocol are located.
14. What is the role of ANVISA in this process?
ANVISA (Agência Nacional de Vigilância Sanitária) is the regulatory body. They must approve the “Compassionate Use” request. If they deny it or delay it excessively, the Judiciary can overrule the decision to protect the patient’s right to health.
15. Is a visa required for medical treatment?
Yes, you should apply for a Temporary Visa (VIVIS) for medical treatment. Your Brazilian lawyer and sponsoring doctor can provide the necessary invitation letters to facilitate the visa process at the Brazilian Consulate.
16. Can Polylaminin be shipped to my country?
No. Because it is an experimental substance requiring specific temperature controls and surgical application, it cannot be exported for personal use. The treatment must be performed in Brazil.
17. What happens if the trial window (72h) has passed?
You move from the “Clinical Trial” category to the “Compassionate Use” category. This allows for a more flexible timeframe, focusing on the humanitarian need rather than strict trial protocols.
18. Why is Polylaminin considered better than stem cell therapy?
While stem cells introduce new cells, Polylaminin creates the “bridge” (extracellular matrix) that allows the patient’s own existing neurons to regrow. Many doctors believe this “scaffolding” approach is more effective for structural spinal repair.
19. Can I sue the Brazilian State to receive the drug for free?
As a foreigner, this is legally complex. While possible in theory, it is much faster and more successful to sue for the right to purchase/access the drug rather than demanding the Brazilian taxpayer cover the full costs of a non-resident’s elective treatment.
20. How do I find a qualified lawyer for this?
You need an attorney specializing in Health Law (Direito Médico) with experience in ANVISA regulations and international litigation. Our firm specializes in bridging this gap for patients worldwide.
Author:

Vinícius Machado | OAB/SP 411.228
Specialist Attorney in Health Law and Founder of the Health Law Observatory. Combining legal expertise with hands-on experience in Hospital Pharmacy and Hospital Nutrition, he offers a unique scientific perspective on high-cost medication litigation. Education: LL.B. from UNIMEP; Post-graduate from PUCRS; currently pursuing a Master’s degree. Expertise: Specialized in oncology treatment denials, surgical litigation, and advocacy for neurodiversity (ASD and Cerebral Palsy). Leadership: Vice-President of the Blue Angel Association (Associação Anjo Azul) and Member of the Health Law Commission of the Ordem dos Advogados do Brasil (OAB - Piracicaba).